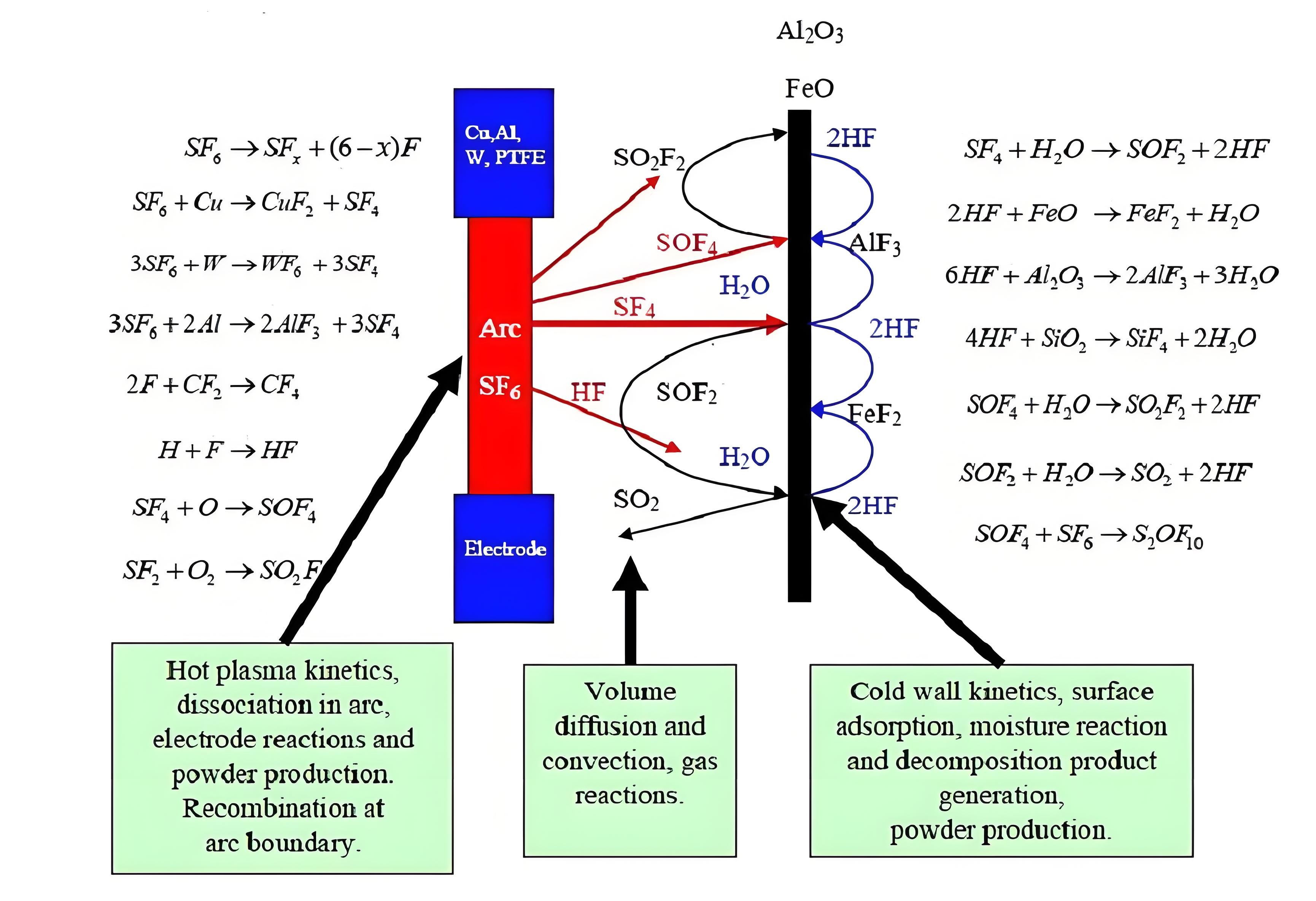
During the switching of normal load or short-circuit current, SF₆ molecules are ionized and fragmented by the arc. The figure illustrates the main reaction processes and the locations where each process is likely to occur. SF₄, as the primary decomposition product resulting from the electric discharge, first reacts with H₂O on the inner wall surface, giving rise to SOF₂. The metal fluorides remain in the form of powder or dust on the surface. H₂O is released in this reaction and thus becomes available for further reactions with SF₄ or for the much slower conversion of SOF₂ to SO₂. In fact, in this process, H₂O is not consumed but rather acts as a catalyst. The factors involved are as follows:
- The formation rate of decomposition products as a function of the arc or discharge energy.
- The transport speed (diffusion or convection) of decomposition products from the arc or discharge location to the walls of the compartment.
- The gas mixing of decomposition products generated at the surfaces (diffusion or convection).
- The reaction rate of decomposition products at the wall surfaces.
- The adsorption rate of decomposition products in the adsorbent materials, which depends on the properties and location of the adsorbent materials.

